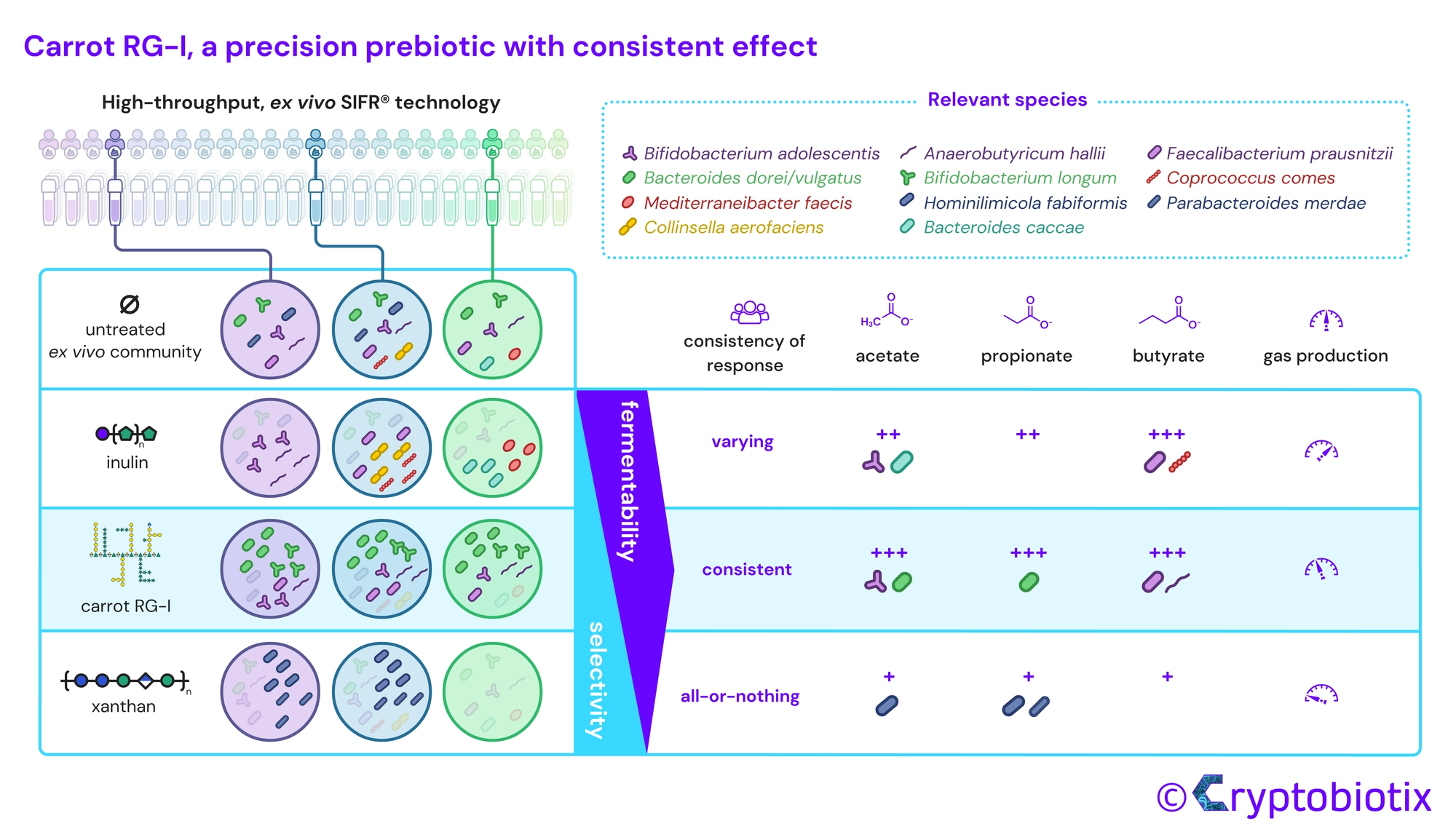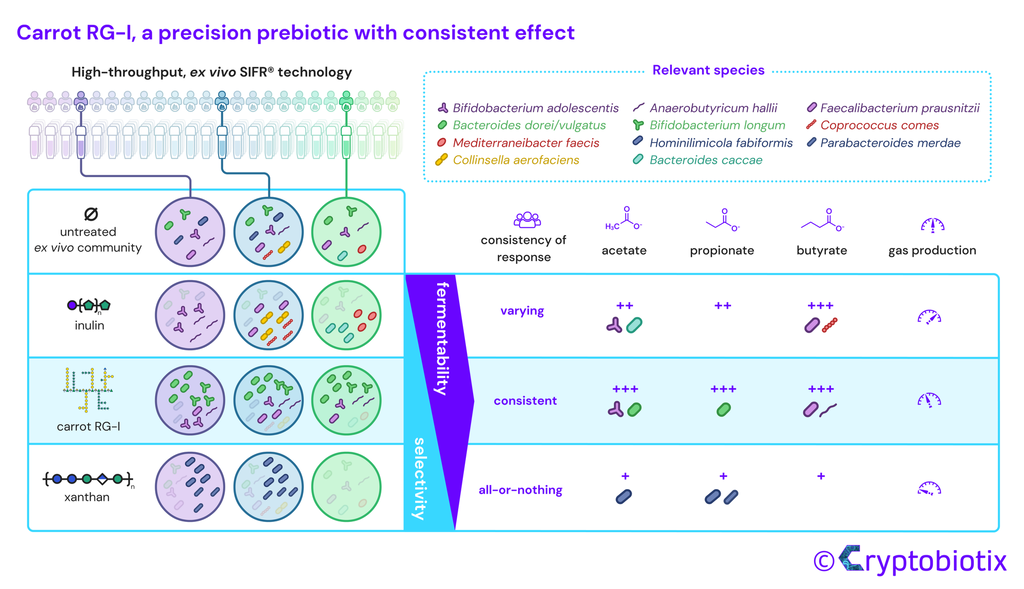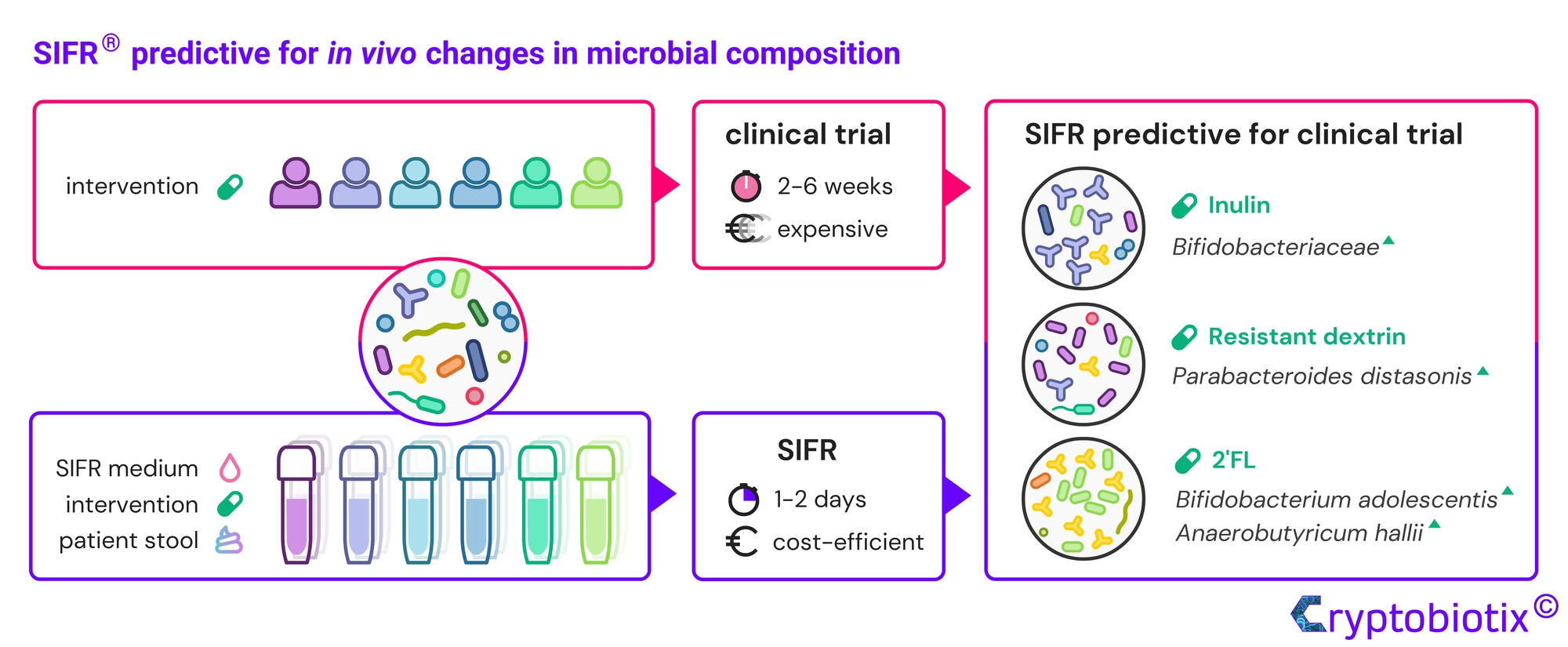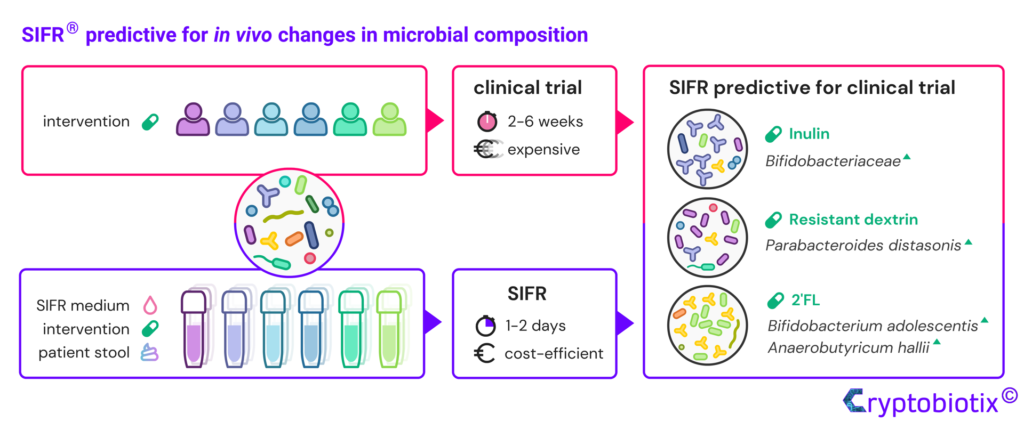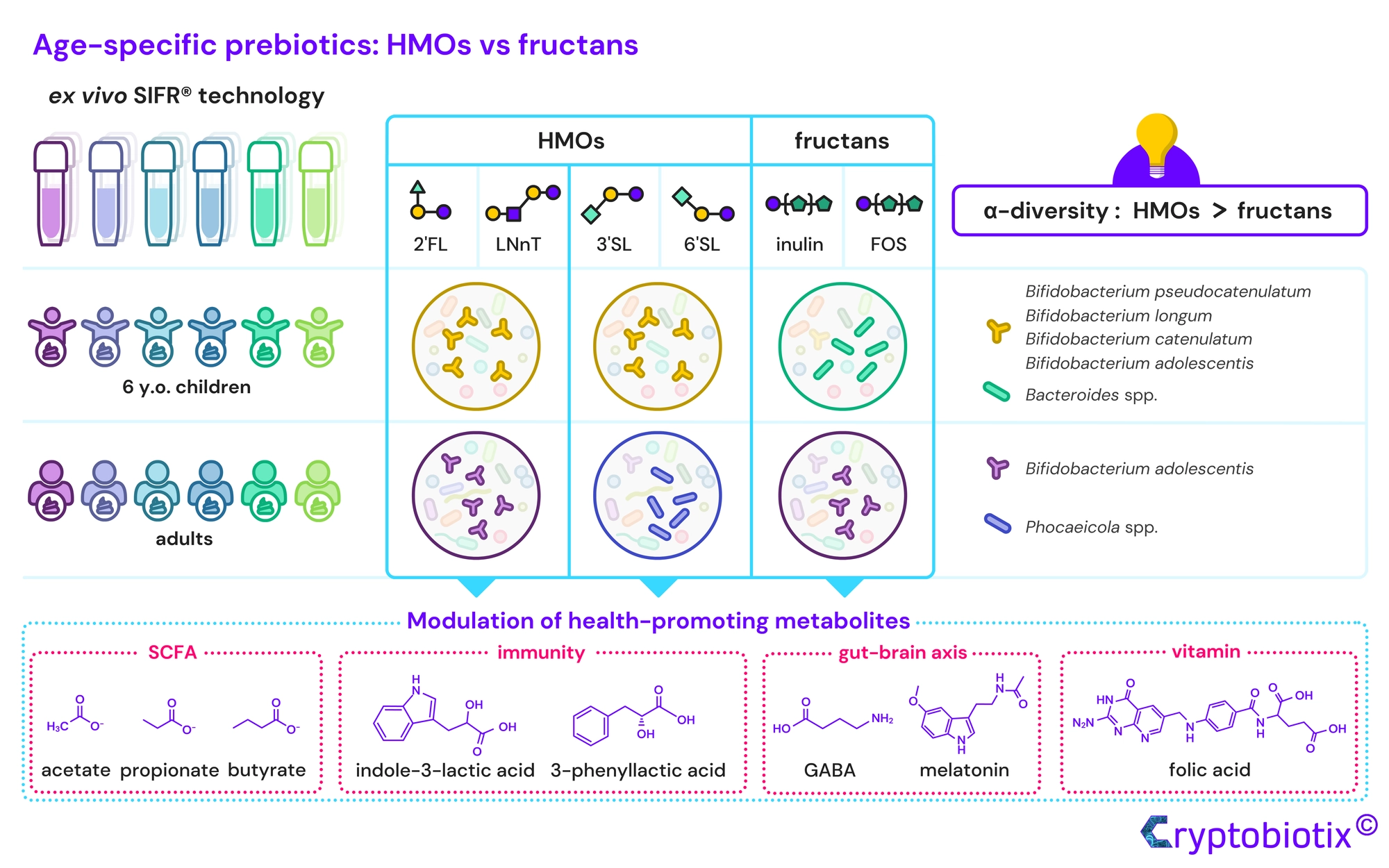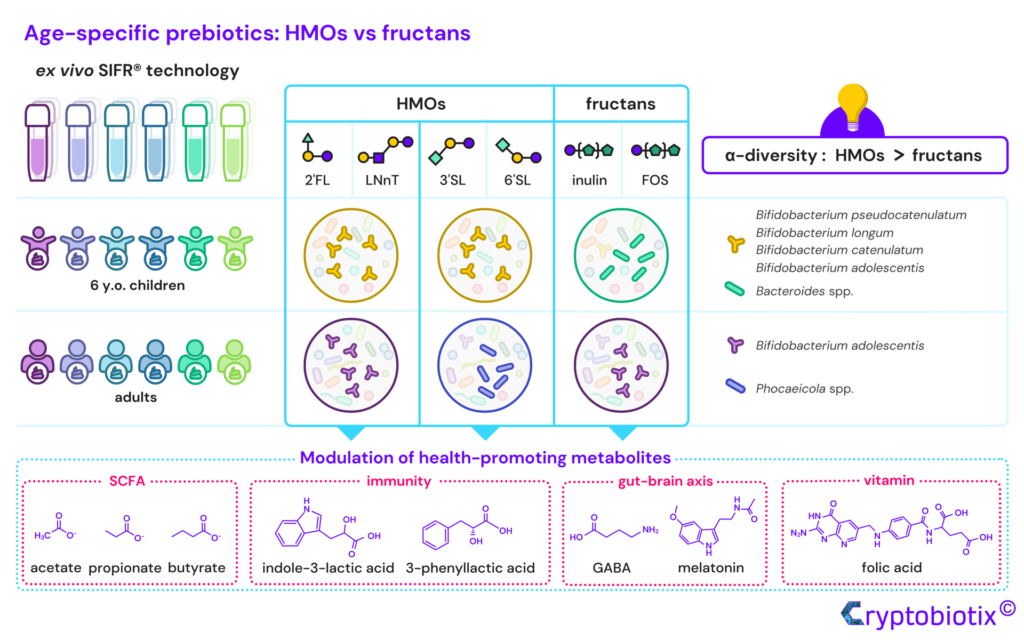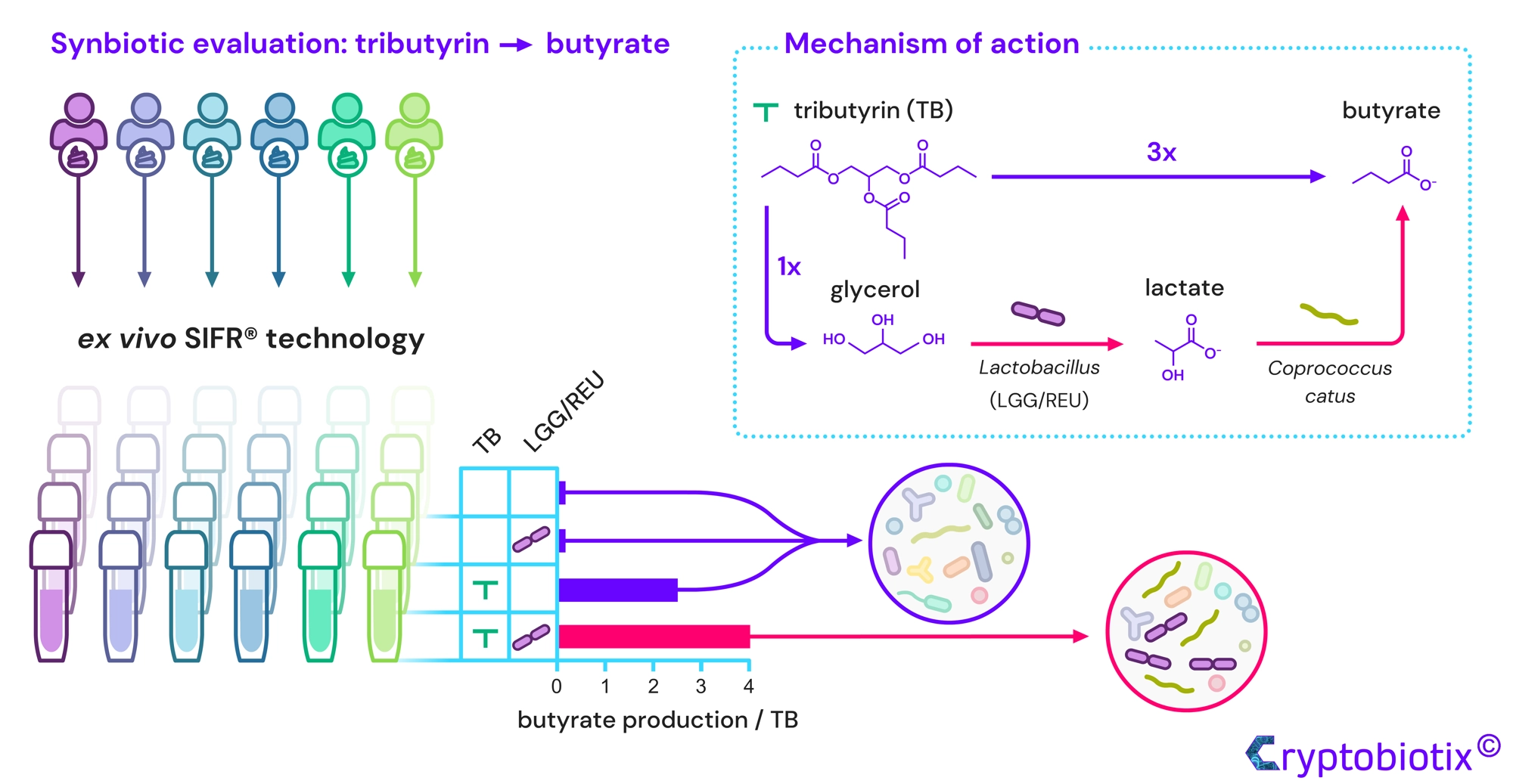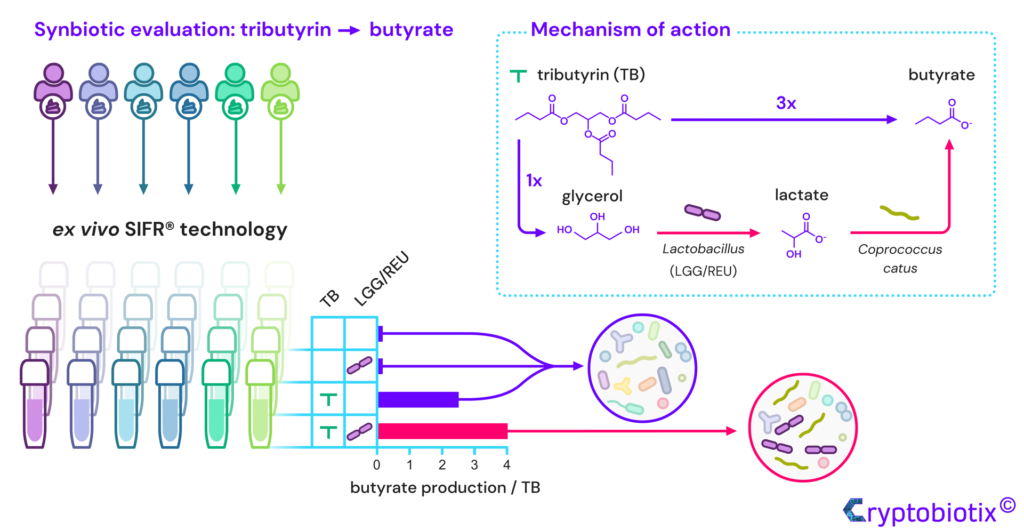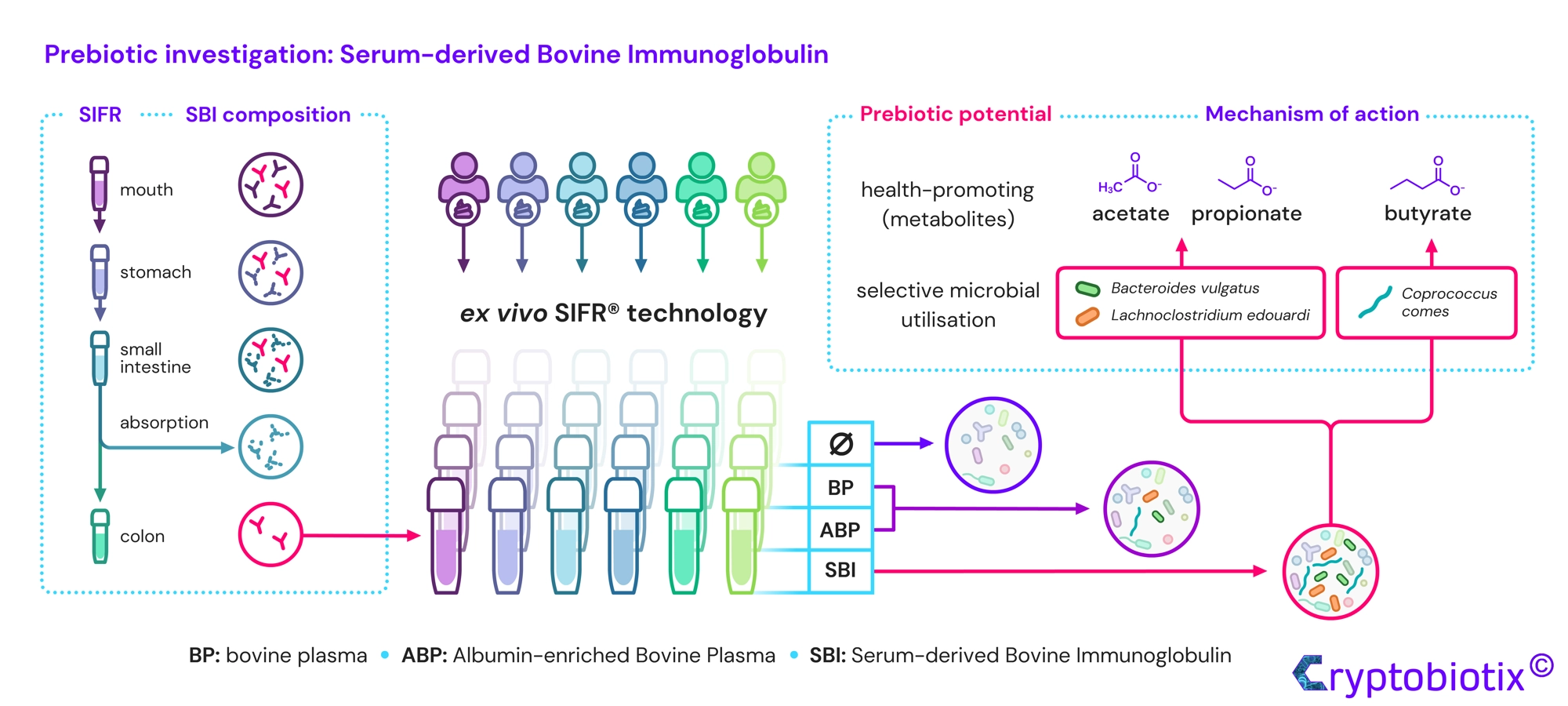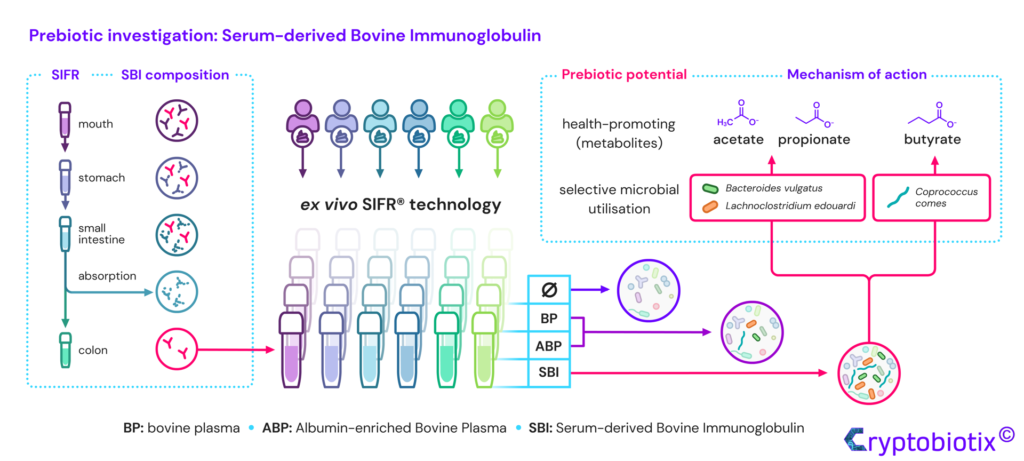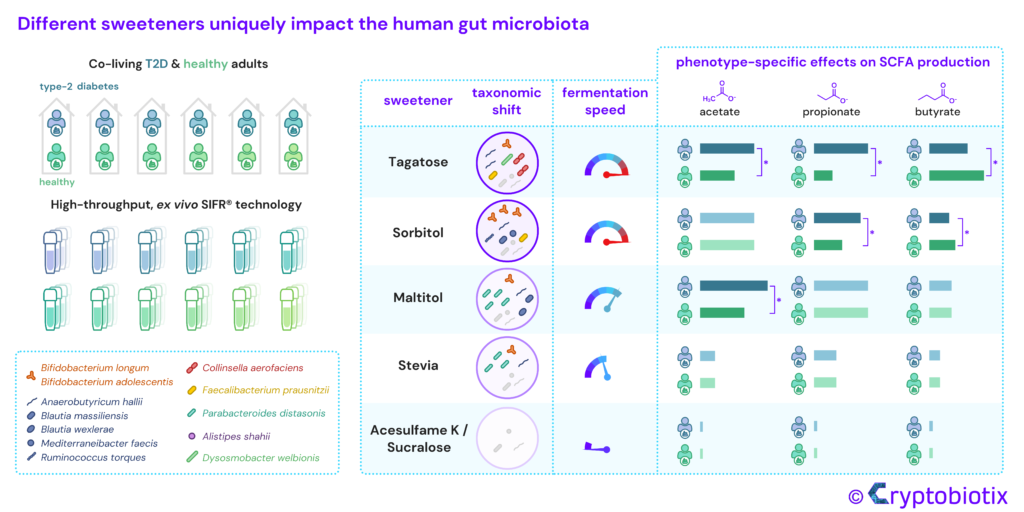
Van den Abbeele et al, 2023, International Journal of Food Sciences and Nutrition
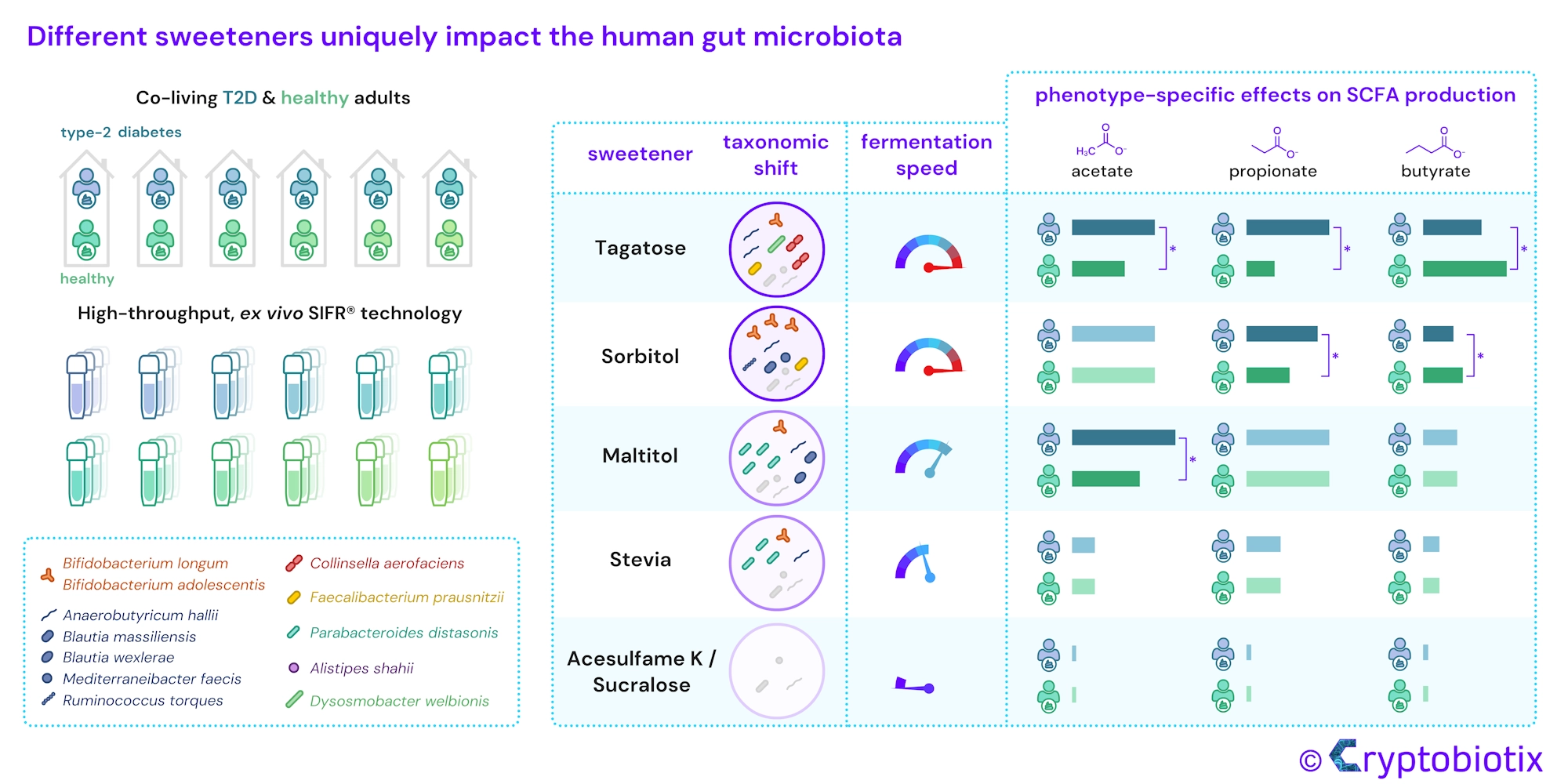
Chronic diseases associated with high sugar intake, such as type 2 diabetes (T2D), impact the microbiome. Low/no-calorie sweeteners are frequently used as sugar substitutes by T2D individuals, and could help in this case, but their effect on the gut microbiota has not been comprehensively studied. We conducted a preclinical research project to help shed light on the matter, using the high-throughput, ex vivo SIFR® technology, which is known for its uniquely validated ability to provide predictive insights into clinical outcomes. Our study extensively compared the impact of a broad range of sweeteners (tagatose, sorbitol, maltitol, stevia, acesulfame K and sucralose), each exhibiting distinct fermentation kinetics, on the gut microbiota of 12 co-living healthy/T2D individuals. The aim of choosing those 6 dyads, was to minimise the influence of long-term diet variations. Sweeteners like sucralose and acesulfame K did not impact microbial composition and metabolite production. In contrast, the rapidly fermented tagatose elevated propionate levels in T2D subjects, while increasing butyrate levels in healthy subjects. In addition, sorbitol strongly increased acetate (associated with Bifidobacterium adolescentis) and maltitol strongly enhanced propionate production (linked to Parabacteroides distasonis) in both donor groups. These findings indicate highly product-specific effects of the investigated sweeteners on the gut microbiota.