Decipher the gut
Redefining preclinical digestion & gut microbiome research
The human gut is home to trillions of microbes that collectively form the gut microbiome and play a crucial role in our overall health and well-being. It is important to understand the mechanism of action of gut health modulators, like prebiotics, probiotics & LBP’s, drugs, and many others. Laboratory testing can help, but traditional in vitro gastrointestinal research suffers from many limitations:
- Limited translation of in vitro outcomes to clinical success
- Poor inclusion of inter-individual variation decreases clinical success rate
- Limited throughput and high cost limits data generation
- Limited data generation and interpretation slows down discovery and product development
At Cryptobiotix, we thought: why not combine throughput and predictivity, working ex vivo? We thrive on redefining established processes, both in the lab and in thinking. Challenge us with your research question and let us customise the most suitable path forward in your early and late R&D stages of product development.
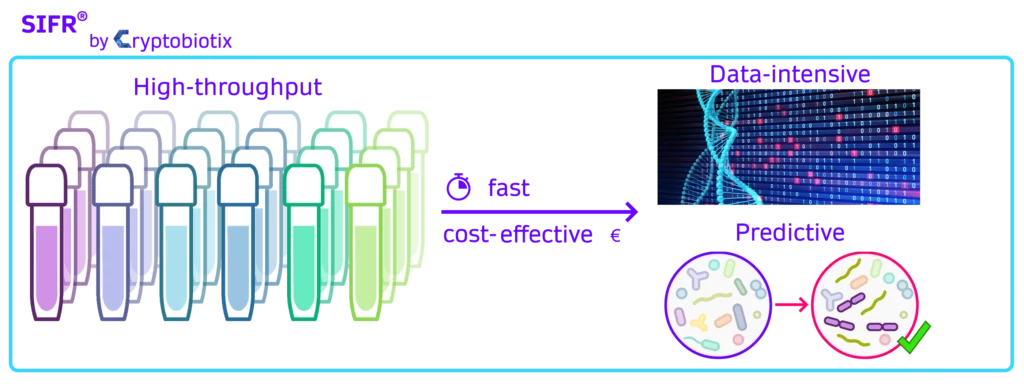
How to generate actionable preclinical insights?
There are three key aspects in how we work:
- Increase biorelevance, translating into predictivity
- Increase throughput by miniaturisation
- Clarify the key findings through advanced data interpretation





What do we do?
Cryptobiotix is a CRO in the gastrointestinal field that provides state-of-the-art preclinical research services using the SIFR® (“cipher”) technology. The name Cryptobiotix refers to the deciphering of the black box that is the gut microbiome, and is derived from the words cryptanalysis and microbiota. We combine in vitro and ex vivo approaches to enable you to investigate more products, in a statistically robust design that considers biological variation.
We answer research questions pertaining to:
- upper gastrointestinal digestion and absorption
- interplay with the gut microbiota and host-microbiome interactions
We use biorelevant, high-throughput models to generate an unprecedented amount of data, that we analyse, interpret and report.
Your applications
Research objective
- Screening & ranking of candidates before clinical trials
- In-depth characterisation of promising lead product
- Generation and protection of IP
- Support your claims
Product type

Functional food

Therapeutics
Simulated host

Our technology: SIFR®
Relying on the latest published insights in digestion and gut microbiome research, the SIFR technology addresses various questions pertaining to digestion and fermentation. Complementing predictivity with operational standardisation, this high-throughput technology allows you to allocate your preclinical budget optimally from early candidate screening to in-depth lead products characterisation.